What is the Difference Between Shape and Geometry of a Molecule? 
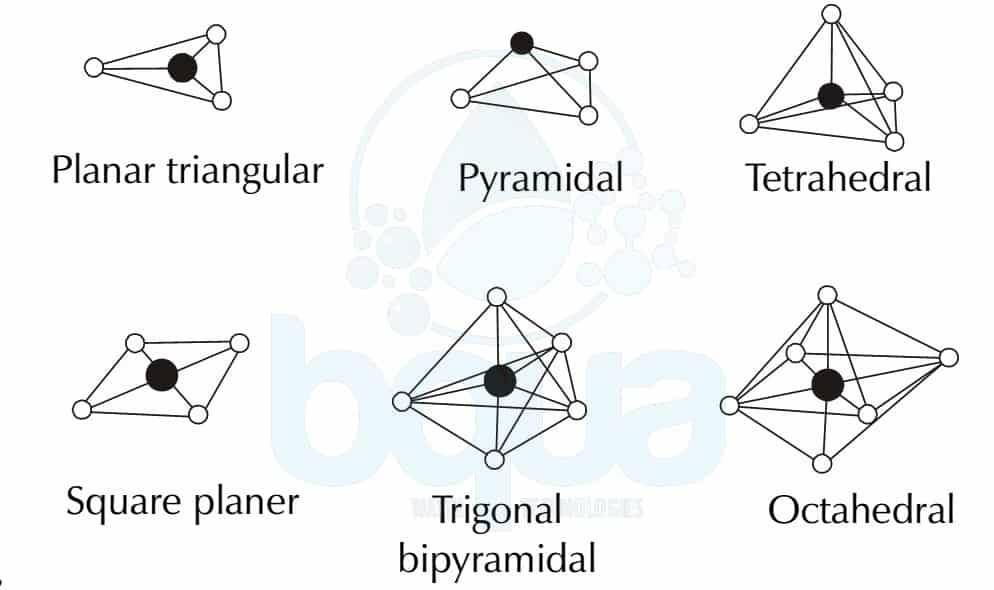
Therefore, the geometry of BeCl2 molecule is linear. Therefore, the number of electron pairs around the Be atom = 4 / 2 = 2 Therefore, the total number of electrons around the central atom = 2 (from Be) + 1×2 (from cl atoms) = 4 For example, the shape of beryllium chloride molecule is predicted as follows: There are three types of repulsion present in between these electron pairs bond pair – lone pair repulsion, bond pair-bond pair repulsion, and lone pair- lone pair repulsion. Here, we can find electron pairs in two types as bond pairs and lone pairs. The basis of this theory is the repulsions between electron pairs in the valence shell of atoms. We can use this VSEPR model to propose a spatial arrangement for molecules having covalent bonds or coordination bonds. VSEPR model is the theory that determines the shape and geometry of a molecule. The shape of the molecule can be predicted using the VSEPR model (valence shell electron pair repulsion model). 
In other words, the shape of a molecule is determined excluding the lone electron pairs of the central atom. The shape of a molecule is the structure of the molecule predicted using the bond electron pair on the central atom. Side by Side Comparison – Shape vs Geometry of a Molecule in Tabular Form However, these are two different terms for some molecules we know. We usually use the terms – shape and geometry of a molecule – interchangeably. The key difference between shape and geometry of a molecule is that shape of a molecule is the structure of the molecule, excluding the lone pair on the central atom, whereas the geometry of a molecule describes the arrangement of lone pair and bond pair electrons around the central atom of the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
